|
Agreement Between
Direct Smear Microscopy and GeneXpert MTBIRIFI
in Diagnosis of Pulmonary Tuberculosis Among Patients
Attending Consultary TB Clinic, Kirkuk
......................................................................................................................................................................
Bushra Mohammad Mahmoud (1)
Suheila Shamse Elden Tahir (2)
Mohammed Abdul-Aziz Kadir (3)
(1) Bushra Mohammad Mahmoud, MS.c, Microbiology,
Consultary Clinic for Chest and Respiratory Disease,
Kirkuk, Iraq.
(2) Suheila Shamse Elden Tahir, MS.c , Family
& Community Medicine,
Consultary Clinic for Chest and Respiratory Disease,
Kirkuk, Iraq
(3) Mohammed Abdul-Aziz Kadir, Ph.D., College
of Medicine,
Kirkuk University, Kirkuk, Iraq.
Correspondence:
Bushra Mohammad Mahmoud, MS.c, Microbiology,
Consultary Clinic for Chest and Respiratory Disease,
Kirkuk
Iraq
Email: gkirkuk@gmail.com
|
ABSTRACT
Background: Tuberculosis is one of
the deadliest diseases causing morbidity
and mortality worldwide, as it infects 9
million and kills 3 million annually, yet
one third of the world population is infected
with TB. This study was planned to compare
the accuracy of PCR with direct sputum smear
among patients who attended the TB Clinic
in Kirkuk.
Patients and Methods: A cross sectional
study recruited 958 patients who presented
to chest and respiratory diseases clinic
for the period from 4th of December till
30th June 2014 for detection of Mycobacterium
tuberculosis by direct smear microscopy.
Out of these 321 samples were tested by
gene Xpert; they were stratified according
to site of disease to 249 pulmonary and
72 extra pulmonary cases.
Results: It was found that the percentage
of direct smear positive was 3.03% while
that for PCR was 11.5%. The percentage of
Rifampin resistance was (10.8%) among positive
cases.
Conclusions: It is concluded that
GeneXpert/MTB/RIF is superior to direct
AFB in detecting Mycobacterium tuberculosis,
by time and detecting resistance in less
than 2 hours.
Key words: Direct smear microscopy,
GeneXpert, pulmonary tuberculosis
|
Tuberculosis is one of the most
deadly diseases worldwide; about 2 billion people
in the world carry latent TB; more than nine million
will become ill and about three million will die
(1).
Poor health system, limited laboratory capacity
for case detection, treatment barriers and complications
make TB a major challenge for public health programs
(2).
Detection of Mycobacterium species is mainly detected
by Ziehl-Neelson stain which employs acid-fast
staining technique. First the sample is inversed
in hot carbolfuchsin that stains red, then the
sample is stained with acid-alcohol and counter
stained with methylene blue. On examination, the
bacteria appear bright red against a blue background
(3).
The Xpert MTB/RIF assay is a fully automated molecular
diagnostic test for TB disease, developed in partnership
with Cepheid Inc, the foundation for innovative
new diagnostics (FIND); the university of medicine
and dentistry of New Jersey (UMDNJ) and the national
institutes of health (NIH). It can detect rifampin
resistance from sputum specimens in less than
2 hours and minimizes staff manipulation and biosafety
risk (4).
The study was conducted to:
1. Show the agreement of conventional and
new methods in detecting Mycobacterium in sputum
and other body fluids.
2. To detect rifampin resistance among
smear positive cases.
A cross sectional study recruited
72 patients who presented to Chest and Respiratory
Disease Clinic in Kirkuk for the period between
4.12.2013-4.2.2014. Sputum samples were obtained
from each patient for direct smear microscopy
and GeneExpert for detection of Mycobacterium
tuberculosis, then agreement in results of both
investigations was assessed.
Regarding sputum sampling each TB suspect was
requested to submit two sputum specimens, one
for conventional smear plus Xpert MTB/RIF and
the second for the conventional smear alone. When
the results of Xpert were positive the Rifampin
sensitive patients were registered as Cat 1 or
Cat 2 according to drug history. If the specimen
proved to be RIF/ resistant, it would be subjected
to culture and drug sensitivity test (DST).
Table 1 shows that during the
period of the study; a total of 958 sputum samples
were examined for direct smear microscopy to detect
the presence of mycobacterium tuberculosis.
Regarding GeneXpert (PCR), a total of 321 were
examined; all smear positive cases, highly suspected
smear negative TB cases and other body fluids
referred by public and private sectors were included
in the study.
Regarding gender difference, the ratio of male
to female was 1.9:1. The percentage of positive
cases by DSM was 3.03%, while in PCR it was 11.52%.
It was clear that there was significant statistical
difference between the two tests (P<0.05).
Table 1: Distribution of cases in DSM &
PCR according to Gender
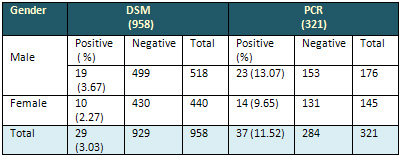
It is clear that PCR has detected higher positive
cases than the conventional method AFB; also it
is obvious that males are affected more than females.
It was found that the proportion of positive cases
in GeneXpert was greater than that with AFB. There
was a statistically significant difference between
the two methods (P< 0.007), as indicated in
Table 2.
Table 2: Agreement between
DSM and GeneXpert for study sample
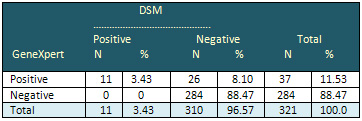
Table 3 illustrates the
distribution of positive cases detected by PCR.
Out of 321 samples tested for detection of MTB
by GeneXpert, 37 cases were positive, distributed
as 35 pulmonary TB and 2 extra pulmonary (pus);
apart from these 2 extra pulmonary samples , all
other samples were negative.
Among the 37 positive cases only 4 (10.81%) cases
were proved to be resistant to Rifadin by the
new test (GeneXpert). The percentage of Rifadin
resistance was 11.42% among pulmonary cases.
Table 3: Distribution
of types of TB cases according to gender and Rifadin
resistance detected by PCR
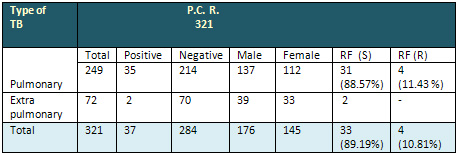
RFS=Rifadin sensitive; RFR=Rifadin resistant
It is obvious from table
3 that PCR has advantage to detect Rifampin resistance
cases within less than 2 hours.
Regarding other body fluids, all were negative
when tested by PCR.
WHO have started the largest
roll-out of GeneXpert, as detecting TB rapidly
and identifying drug resistance on the spot is
an essential step to improve care of the infected
people and avoid transmission in the community
(5).
The higher percentage of positive cases among
males than females detected by PCR, is in agreement
with WHO report as males have much higher rates
than females.
In a study conducted in Vantanilla, Lima, in 2005
showed that tuberculosis globally kills more women
than any other single infectious disease. It is
noted that 70% of the poor are women and they
face obstacles in seeking medical advice and care
(6).
Detection of Mycobacterium TB by PCR was increased
in comparison with direct sputum smear; this result
is in agreement with a study done in Brazil and
published as Clinical trials during 2011.
In a survey conducted among prisoners, in Malaysia,
it was documented that a single GeneXpert assay
proved to get 53% sensitivity, 100% specificity
and concluded that single test yields low screening
sensitivity (7).
WHO technical report has concluded that PCR is
the most common method of amplification, in spite
of many other methods for rapid test for diagnosing
MTB which are commercially available as in Roche
(Amplicor PCR-FDA-approved), Becton Dickson CBD
probe Tec, Standard Displacement amplification
(SDA), Gene probe (Amplified Mycobacterium tuberculosis
Direct (AMTD),Transcription machinated amplification
(TMA) in addition to the assay used in the current
study under the commercial name Cepheid (GeneXpert
MTB/RIF) nested real time PCR (8).
In addition to excellent sensitivity with smear
positive specimens, the device can identify 72.5%
of smear negative culture positive samples from
single tests. WHO recommended that the GeneXpert
MTB/RIF is suitable for use at district and sub
-district levels (9&10).
Detection of drug resistance in comparing the
conventional (AFB) test and new PCR, showed that
males had higher resistance than females although
the number of cases is small as tuberculosis is
a rare disease and among all notified cases resistance
occupies 3% of the TB cases (11).
In MTB, mono resistance to (RIF) is rare and almost
90% of resistant cases to RIF are also resistant
to INH so a positive result of RIF is a strong
surrogate of MDR-TB, and in resistant isolates
it has been shown that from to 95% to 98% RIF
resistance is caused by mutations in the proB
gene encoding the RNA polymerase B-unit (12&13).
1-Colledge, N. R.; Walker, B.R.
& Ralston, S.H. (2010). Davidson's Principles
& Practice of Medicine. 21 st Edition. Churchill
Livingstone Elsevier, Edinburgh.
2-Maiga, M; Mohoudan, A.; William, R. & Bishai,
et al (2012). Current tuberculosis diagnostic
tools role of urea breath test.
Indian J. Med. Res., 135: 731-736.
3-Virella, G. (2004). Microbiology and Infectious
Diseases. 3rd Edit. Williams & Wilkins PP.
168-174.
4-Piatek, S.A.; Cleff, M.V.; Alexander, H. et
al. (2013). Gene expert for tuberculosis diagnosis
planned and purposeful implementation.
Global Health Sci. Pract., 1, 18-23. WWW. Ghsc
Journal.org.
5-Onifade, D.A.; Bayer, A.M.; Montaya, R. et al.
(2010). Gender related factors influencing tuberculosis
control in Shanty towns. A qualitative study.
BMC Public Health, 10, 381.
6-Keertan, D. (2013) Trial of point-of-treatment
Xpert MTB/RIF Assay.
Clin. Trials. Gov. Identifier. University of Cape
Town.
7-AbduRazzak, H.; Al-darragi, A.; Razak, H.A.,
et al. (2013). The diagnostic performance of a
single Genexpert MTB/RIF Assay in an intensified
tuberculosis case finding among HIV infected prisoners
in Malaysia.
PlosONE 8(9).
8-WHO (2011) Automated Real-time Nucleic acid
Amplification Technology and Rapid and Simultaneous
Detection of Tuberculosis and Rifampin Resistance.
Xpert MTB/RIF system. Policy statement.
9-UNITAID (2012) Tuberculosis, Diagnostic Technology
Landscape Semi-annual update.
10-Merza, M.A.; Famia, P.; Tabars, P. et al. (2011)
Anti Tuberculosis drug resistance and associated
risk factors in a tertiary level TB Center in
Iran.
A retrospective analysis. J. Infect. Dev. Ctries,
5(70: 511-519.
11-Zeka, N.A.; Tasbakan, S.; Cavasogli, V.C. (2011).
Evaluation of the Gene Xpert MTB/RIF. Assay for
rapid diagnosis of tuberculosis and detection
of Rifampin Resistance in Pulmonary and Extra-pulmonary
specimens.
J. Clin. Microbiol, 49(12): 4138-4141.
12-Prachi Pinglay (2012) GeneXpert test for drug
resistant TB detect 28 cases during month. Hindustan
times, Mumbai, April 28.
13-Osama, M.; Simpsa, J.A.; Galdwell,j. et al.
(2014) GeneExpert MTB/RIF Version G4 for identification
of Rifampin-Resistance Tuberculosis in a programmatic
setting. J. Clin. Microbiol., 52(2): 635-637.
|
