|
Biological markers
study of acute and chronic myeloid leukemia
......................................................................................................................................................................
Azhar Abbas
Ismaeel
Correspondence:
Azhar Abbas Ismaeel
College of Education for pure Science, Ibn Al-Haitham,
Department of Chemistry, Biochemistry Laboratory,
University of Baghdad, Iraq
Email: Azhar.almula@yahoo.com
|
ABSTRACT
Twenty two acute myelogenous leukemia (AML)
patients and 20 chronic myelogenous leukemia
(CML) patients, were admitted to Medical
City Hospital, in addition to 35 healthy
individuals as the control group. Vitamin
C and vitamin D, trace and essential elements
cobalt, and Nickel magnesium were evaluated
in the sera of all above individuals, using
high performance liquid chromatography and
atomic absorption spectrophotometry for
evaluating vitamins and elements. The results
revealed a a significant decrease in vitamin
C and vitamin D and Mg levels in both patients
groups compared to controls and a significant
difference in CO and Ni levels in sera of
AML and CML was found.
Conclusion: Vitamins, trace and essential
elements abnormalities occur in both patient
groups compared to Controls.
Key words : Acute, Chronic leukemia,
vitamins C, D, cobalt, Nickel, Magnesium
|
Leukemia is a type of cancer
of the blood or bone marrow characterized by an
abnormal increase of immature white blood cells
called "blasts. It is classified into several
types. Acute and chronic could be considered as
the essential type and includes many types under
each term such as lymphocytic, granulocytic and
monocytic leukemia((1). Acute leukemia, is one
of the two major categories of leukemia, with
symptoms including anemia, fatigue, weight loss,
easy bruising, thrombocytopenia, and granulocytopenia
,while chronic leukemia is characterized by the
excessive build up of relatively mature, but still
abnormal, white blood cells (2). The maintenance
of optimal health proteins, lipids, macronutrients,
micronutrients, vitamins and trace elements.(3)
Vitamins are an organic compound required by an
organism as a vital nutrient. In limited amounts
,vitamins serve multiple roles and they work together
to perform many functions, from reducing infection
to aiding metabolism. (4) Vitamin C (Ascorbic
Acid) is an important antioxidant found in the
biological system. It is the first line antioxidant
defense in plasma.( 5) Proposed mechanisms of
vitamin C activity in the prevention and treatment
of cancer include : enhancement of the immune
system by increased lymphocyte production, stimulation
of collagen formation necessary for "walling
off" tumors , inhibition of hyaluronidase,
keeping the ground substance around the tumor
intact and preventing metastasis, inhibition of
oncogenic viruses, correction of an ascorbic deficiency,
and is often seen in cancer patients.(6,7,8)
Cholecalciferol is a prohormone
that is synthesized in the skin by photochemical
conversion of 7-dehydrocholesterol. It is subsequently
hydroxylated to 25-hydroxycholecalciferol [25(OH)D3]
in the liver and in the kidney finally to the
active metabolite, 1,25 didydroxcholecalciferol
[1,25(OH)2D3. ( 9 ,10)
Trace elements (TE) are a group
of elements that are present in the human body
in very small amounts (milligrams per day), but
are nonetheless important to good health. ( 11)Their
action is that (TE) are constituents of, or interact
with enzymes, cofactors, phosthetic groups or
hormones that regulate the metabolism of much
larger amounts of biochemical substrates(12 ).
Cobalt is an essential element
for humana, as a part of vitamin B12 (coblamin)
- micro flora of the lumen intestine.1(13,14,15,)
Nickel is one of the ultra trace elements; Nickel
ions used in the iron transport system across
small intestine related with ferritin, thereby
cause anemia;the deficiency of nickel causes alteration
of folate and vitamin C.(16)
Magnesium (Mg) is essential to the basic nucleic
acid chemistry of life and thus is essential to
all known living organisms. Mg is important for
maintaining of DNA. Magnesium is extremely necessary
for proper ATP synthesis(17,18,19).
The aim of the study is to evaluate
vitamin C, vitamin D and trace and essential elements
of Co, Ni, Mg, levels in the sera of patients
with acute and chronic leukemia compared to controls.
- Experimental Part
- Subjects:
Blood samples were collected from the patients
with acute myelogenous leukemia (AML) and patients
with chronic myeloblastic leukemia (CML) from
men, who were admitted to Medical City Hospital.
Their age range was 32-52 years. The patients
were diagnosed by specialist doctors. Thirty five
healthy individuals were matched aged 30-50.
Instrumentation : Vitamin C was measured
using high performance liquid chromatography (
Shimadzu Japan C18 column). Vitamin D was separated
on FLC (Fast liquid chromatograph column) 3µm
particle size stored column, obtaining optimum
conditions, temperature, flow rate, eluent composition
were used for detection of vitamins . The concentration
of each vitamin was estimated through measurement
of area under the curve of chromatograms of standard
vitamins and for patients and control serum samples.
Cstandard = Standard concentration
of the measured vitamin in ug/ml.
Vitamin concentration in ug/ml
= Asample x Cstandard
...................................................Astandard
Asample = Area under the
curve of sample.
Astandard = Area under the
curve of standard
Atomic absorption spectrophotometry (model
AA46-schma-dzo-japan{AAS) : AAS was used for
determination of CO, Ni and Mg. A standard calibration
curve is used for measurement of CO, Ni and Mg
in the sera of groups under this study AML, CML,
and controls.
Statistical analysis
The means and standard deviations, and student
t test was used to compare the significance of
the difference in the mean values of any two groups.
(P < 0.05) was considered statistically
significant (20).
Table 1 shows low serum levels
of vitamin C (3.06, 3.46 µg|ml) for AML,
ACML compared with controls (4.2µg|ml).
It was reported that toxic reactive oxygen species
increase during leukemia(21). Ascorbate is a powerful
reducing agent capable of rapidly scavenging a
number of reactive oxygen species, so our results
are in agreement with results which illustrated
that the cancer incidence and mortality is associated
with low levels of ascorbic acid.(22) We can speculate
that this protective effect may occur through
antioxidant pathways or through enhancement of
immune function. (21) There is also some recent
evidence suggesting that ascorbate exerts a selective
cytotoxic effect against malignant cells; all
these observations indicate
that supplemental ascorbate should be of some
therapeutic value in the treatment of cancer.(
22)
Table 1 shows levels of vitamin
D (3.46, 3.63 µg|ml) for AML, ACML compared
with controls (4.73µg|ml). The reduction
of vitamin D level in AML and CML is in agreement
with Drake, MT, Maurer MJ(23). Numerous epidemiological
and preclinical studies support a role of vitamin
D compounds in cancer prevention and treatment
in colorectal, breast, prostate, ovarian, bladder,
lung and skin cancers and leukemia(24 ).The newly
recognized use of 1,25(OH)2D3 analogues as immunomodulatory
agents is based on the ability of these analogues
to influence gene expression in cells of the immune
system and cytokine expression by other cells(25).
In addition some non traditional roles ascribed
to vitamin D include anti-flammatory and immune-modulating
effects. (26)
Table 1: Serum concentration (µg|ml )
of vitamin C and D in AML, CML and healthy control
group.
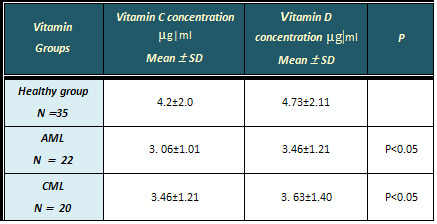
Table 2 shows the results of cobalt, Nickel and
magnesium, levels in sera of controls, AML and
CML groups. The result of cobalt level in serum
of patients with AML, CML, were (0.012 58 , 0.0154
ug/l ), respectively,
which are lower than serum level of controls (
0.027930 ug/l). This
result is in agreement with published results
(0.03 ug/l) (27). It
appears that the depletion of Cobalt in serum
of patients is related to vitamin B12. (15) In
addition, biological research that has been done
suggests a relationship between cobalt and cancer(28).
Table 2: The serum concentration( µg|l)
of CO, Ni and Mg in AML, CML and healthy control
group
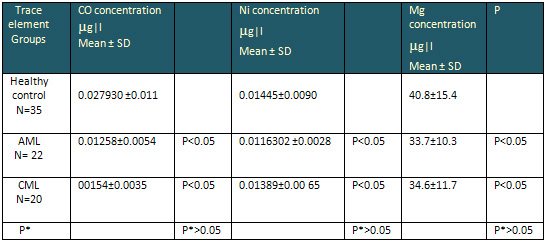 P* value between AML and CML
P* value between AML and CML
The Nickel level in serum of
patients with Acute CML were 0.01163 and 0.01390
respectively, which is lower than level in serum
of controls, ( 0.01445 ug/l).
This result is compared with Nickel level of patients
and controls with published values ( 0.3 ug/l
) (29, 15 ). In another study, high serum concentration
of Nickel causes cancer because itdamages the
chromosomes , and on a molecular basis Nickel
induces DNA damage (DNA strand breaks and crosslink's
infidelity of DNA replication, inhibition of DNA
repair, and the helical of B-DNA to Z-DNA ) by
binding Nickel to the nuclear protein ( 30 ).
These events reflected the effects of Nickel in
two states of concentrations, in the statement
of exposure to and the statement of lower levels,
tended to cause cancer (31).
The Mg level in patient groups is significantly
lower than controls. The decrease in Mg concentration
in the many studies suggests the diminished Mg
due to the high ATP requirement of the leukemia
since Mg is known to be an important part as a
cofactor in most of the energy producing reactions(32).
Figure 1, Figure 2 and Table 3 show the correlation
relation between vitamin D and CO, Ni in sera
of AML, CML and healthy control group; positive
correlation between vitamin D and CO, Ni in healthy
control group with r values (0.987,0.799), positive
correlation between vitamin D and CO in AML and
CML with r values (0.980,0.970); positive correlation
between vitamin D and Ni in AML and CML with r
values (0.948, 0.870)
Table 3: Correlation relation
between Vitamin D and Co, Ni in AML, CML and healthy
control group.
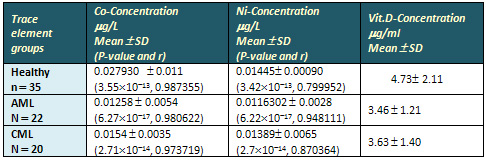
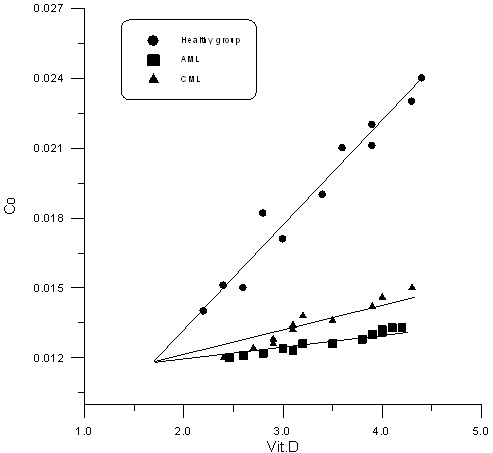
Figure 1
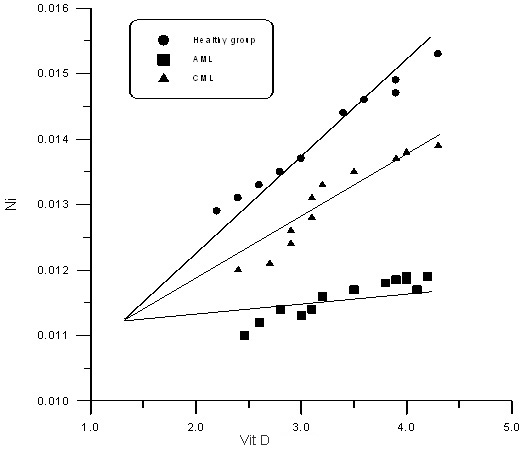
Figure 2
1- Meson, J. N. St C.; Dennis L. Kasper; Harrison,
Tinsley Randolph; Braunwald, Eugene; Fauci, Anthony
S.; Hauser, Stephen L; Longo, Dan L . Harrison's
principles of Internal medicine. New York2005, Medical
Publishing .
2- Colvin G. A., Elfenbein G. J. The latest treatment
advances for acute myelogenous leukemia. Medicine
and Health, Rhode Island 86 (8): 243-6 . . 2003
3- Bender, David A.. Nutritional biochemistry of
the vitamins. Cambridge, U.K.: Cambridge University
Press 2003.
4- Lieberman, S and Bruning, N. The Real Vitamin
& Mineral Book. NY: 1990.
5- Halliwell B. Biochemistry of oxidative stress.
Biochemical Society Transactions, 35, 1147-1150,
2007.
6- Basnet P, Norsk farmaceutisk tidsskrift Vitamin
C prevention of chronic Disease and optimal Doses,
9; 20-24, 2010
7- Kathleen A. Head, ND. Ascorbic Acid in the Prevention
and Treatment of Cancer, 2004
8- Akoury, N.E. Willy High dose Vitamin C is an
Alternative cancer treatment 2012.
9- Binkley, N. , R. Ramamurthy, L ow vitamin D status:
definition, prevalence, 2010.
10- Holick M. Vitamin D: the underappreciated D-lightful
hormone that is important for skeletal and cellular
health. Curr Opin Endocrinol Diabetes;9:87-98 2002
11- Somous N. "Trace elements . in clinical
nutrition : parenteral in medical practice "
, 4th ed , Royal P.M.S,London,P.442 ;1977 nutrition
' 2nd edition. Philadelphia 156 - 183,1993
12- Harrison PM,Arson p. The ferrins; molecular
properties, iron storage function and cellular regulation.
biocembiophy s Acta ; 275 . 161-203,1996
13- Nasri H. Chelation, Downloads, Erythrocyte (Red
Blood Cell) Essential & Toxic Elements, PP.1-3
. 2006
14- Atlanta GA ., Agency for Toxic Substances and
Disease Registry ( ATSDR), P1- 2 . 2001,
15- S. Hosokawa, H. Nishitian , K. Umemura, T .Tomoyoshi
, K. Sawanishi and O. Yoshida , (International Urology
and Nephrology SpringerLink- Journal Article), 447
- 451 .2007.
16- Hopfer SM, Fay WP. Sunderman FW Jr ., Ann .
Clin . Lab. Sci ., , 19 , 161- 167 . 1989
17- Jane Hidon, Ph.D. Linus Pauling Institute Oregon
State University "Magnesium",2003
18- Hartwig A. Role of magnesium in genomic stability
mutate. Res 475:113 - 121 , 2001
19- A.L Buchachenkoa, L.N. Schegolerab and N.N.
Breslavrakyca paramagnetic complex of magnesium
as mediators in enzymetic ATP synthesis, 2009
20- Bailey N.I Statistical methods in biology; Ltd
press.,1974. 20- 0
21- Richterc, Gogvadzev, Laffranchi R, Biochem.
Biophy ra ; Orlando- S-F; Boldt, David H, Br-J-Haematol
- 117: 727 ,2002
22- Mallery DL, McEwan WA, Bidgood SR, Towers GJ,
Johnson CM and James LC. Antibodies mediate intracellular
immunity through tripartite motif containing 16;
107(46):19985-90.21 2010.
23- Drake, M. T., M. J. Maurer, Vitamin D insufficiency.
2010
24- Harrison JS, Bershadskiy A. Leuk Res Treatment.
, 2012 .
25- Holick, M. F. Vitamin D: evolutionary, physiological
and health perspectives." CurrDrug Targets
12 ,14-18. 2011 .
26- Gueli, N., W. Verrusio. Vitamin D: drug of the
future. A new therapeutic. 2011.
27- H. M.Shen and Q.F. Zhang , Environ Health Perspect
, 102 , Suppl 1 , 275-282 . 1994
28- Sheppard, P.R., R.J. Speakman, G. Ridenour,
and M.L. Witten. Temporal variability of tungsten
and cobalt in Fallon, Nevada. Environmental Health
Perspectives 115:715-719. 2007.
29- Evert Nieboer and Jon Oyvind Odland, Physician
copy - Nickel, Oslo, April 11, pp.785 - 796. 2001.
30- J.M. Christensen , J. Kristiansen , N.H. Nielsen,
T. Menne and Byrialsen , Toxylogy Letter, 108, Issues
2-3, 185- 189 ,2000.
31- Swain R., J Fam Pract, 41, 6 , 595- 600 ,1995.
32- J. Durlach, P. Rinjard, M. Bara, A. Guiet-Bara,
and P. Collery, in Magnesium: Physiologische Aspekte
für die Praxis (B. Lasserre, ed.), Panscientia
Verlag, Hedzingen-Zürich, 1987
|
